Table of Contents
Introduction:
“The standard format for submitting submissions, modifications, supplements, and reports to the Center for Drug Evaluation and Research (CDER) and the Center for Biologics Evaluation and Research (CBER) of the FDA is known as electronic Common Technical Document (eCTD).
Understanding the eCTD requirements and applying them effectively to your submissions is critical.
Such specifications are not always sensitive, however, and headaches that occur when documents do not fit seamlessly into the eCTD structure that is permitted.
If that happens, sponsors will face last-minute changes, putting deadlines in trouble. Worse yet, the FDA, which is known as technical rejection, could bounce back the entire submission.
eCTD Structure
In the past, paper submissions were a mode of life for anyone submitting an application to the FDA. Unfortunately, paper submissions are bulky, hard to store, and inefficient to assemble, update, and review. In order to eliminate these issues and speed the review process, the FDA now requires electronic submissions for most of the applications.
Since May 5, 2018, all submissions for the new drug applications (NDAs), Abbreviated NDAs (ANDAs), Biologics License Applications (BLAs), Commercial Investigational New Drug Applications (INDs), and Master Files are required to follow eCTD specifications.
There are several variations to the eCTD, but in its simplest terms, the eCTD is a standardized document structure that enables details to be presented reliably and comprehensively within a request.
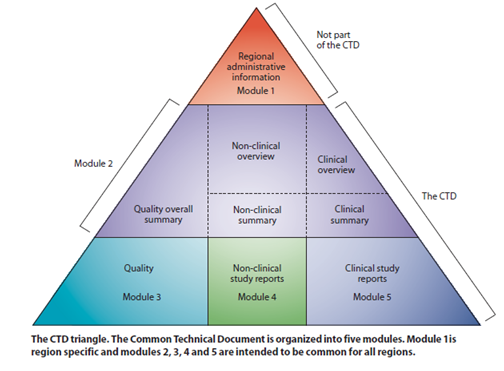
The eCTD submissions comprise 5 sections, called modules, each of which contains a specific type of information.
- Module 1 : (not technically part of the CTD): region-specific administrative information
- Module 2: manufacturing, nonclinical, and clinical overviews and summaries
- Module 3: detailed manufacturing information
- Module 4: nonclinical study reports
- Module 5: clinical study reports
Chemistry, Manufacturing, and Controls (CMC)
As anyone who has written an IND or marketing application knows, the CMC modules (module 3 and summaries in module 2) are generous and can be tedious and time-consuming to complete.
Because more documents usually mean more “cooks in the kitchen,” it’s tempting to combine allowed parts in an attempt to streamline writers and versions management.
Nevertheless, the granularity of the CMC parts should be written with all of the submission’s life cycle in mind.
For instance, if you decide to consolidate module 2.3 (Quality Overall Summary), any updates to the drug substance sections will require the resubmission of this complete module rather than just the pertinent information.
As a general rule, a more granular structure should be measured in order to avoid issues during the lifecycle of the application.
The “M4 Organization of the Common Technical Document for the Registration of Pharmaceuticals for Human Use” can be used as a reference to determine the permitted granularity.”
M4 : The Common Technical Document
“The agreement to assemble all the Quality, Safety and Efficacy information in a common format (called CTD – Common Technical Document ) has revolutionised the regulatory review processes, led to harmonised electronic submission that, in turn, enabled implementation of good review practices.
For industries, it has eliminated the need to reformat the information for submission to the different ICH regulatory authorities.
The CTD is organised into five modules. Module 1 is region specific and Modules 2, 3, 4 and 5 are intended to be common for all regions. In July 2003, the CTD became the mandatory format for new drug applications in the EU and Japan, and the strongly recommended format of choice for NDAs submitted to FDA, United States.
More information: An electronic version of the Common Technical Document (eCTD) can be produced using the information developed by the eCTD Implementation Working Group.”
Clinical Study Reports
“Even the experienced teams can meet problems while determining the best way to structure a clinical study report (CSR) within the eCTD. Should it be submitted as a single document? If splitting it up, what is the proper way to group these sections?
The eCTD structure for Module 5 follows the levels outlined in “The Comprehensive Table of Contents Headings and Hierarchy.” even though legacy CSRs can be submitted as one document, the current recommendation is to divide CSRs into sections: synopsis, report body, and individual appendices (i.e., Section 16 of CSR separated to 3rd level headings).
Preparing a CSR in this format allows reviewers to more simply navigate the large amount of the information, leading to more proficient reviews. Additionally, by dividing the report into sections, changes can be more easily tracked, as the whole report does not need to be replaced if only one section is updated.
Report Numbering
Each study report, both non-clinical and clinical, must have an ID and title of the study inserted into the submission.
As such, it is important to ensure that all reports have an unique id associated with them before they are sent to your publishing team. It usually becomes a concern as data from the study extends over several studies, such as an initial clinical trial and related population PK analysis.
If a population PK analysis is being conducted for a definite clinical study, the population PK report should receive a unique study ID to ensure that the analysis is different from the general clinical study report.
This also helps to ensure that any cross-references in the summary sections are clear as to the content being referenced.
Annual Reports
Back in paper days, the annual report was frequently written as a single document; on the other hand, the current granularity in “The Comprehensive Table of Contents Headings and Hierarchy” outlines multiple sections for annual reports.
While it may be tempting for the sponsors to continue authoring the annual reports as one document, the structure of the eCTD does not readily allow for this approach and can lead to validation errors, putting the application at risk of the technical rejection.
When published as a single text, publishers can seek to avoid validity issues by putting the annual report within a single node that corresponds to one of the parts of the annual report (e.g., Nonclinical Studies Summary).
This method causes ambiguity during the analysis, however, as the content does not fit properly with the definition of the nodes.
Hyperlinking
One of the most popular problems faced by publishers is trying to hyperlink to a section and finding that the section either does not exist or can not be linked to.
Part of this is due to typographic errors during document authoring; however, other instances arise due to the writers’ lack of comprehension about what material could actually be connected to?
As a rule, hyperlinks can only be made to individual documents and not to section folders.
For example, a common mistake when hyperlinking is referencing a section that is of a higher level of granularity than that to which the documents are written.
Some examples include:
- Referencing the module for the single dose toxicity studies (i.e., 4.2.3.1) rather than the specific study
- Referencing module 3.2.P.3 as a whole rather than specific relevant section(s) (e.g., 3.2.P.3.1)
The “Comprehensive Table of Contents Headings and Hierarchy” and “M4 Organization of the Common Technical Document for the Registration of the Pharmaceuticals for Human Use” can be used to determine permitted levels within an application.
In order to make the publishing process as competent as possible, it is important for the writers to ensure that each document has undergone adequate quality control review to verify that all cross-referenced sections exist previous to sending them to the publishing team.”
https://admin.ich.org/sites/default/files/2019-05/CTD_triangle.pdf
https://www.ich.org/page/ctd
https://www.nuventra.com/resources/blog/regulatory-submissions-ectd/
https://www.trilogywriting.com/common-technical-document-ctd/


